
autoloMATE® Launchpad
Contact us If you have any questions about our Terms and Conditions, Privacy Policy or if you would…
08 Jun 2026Life sciences end-to-end digital accelerator














With our extensive audit logging feature, you can have complete trust that everything that happens on the system is tracked & traceable, and every input and change in the execution record is logged with the precise date and time as well as the identity of the recording user.
autoloMATE® is designed to comply with guidance from UK MHRA, EU EMA & US FDA, going a step further through its use of ledger technology to ensure data immutability – records cannot be falsified and this can be demonstrated for every relevant item of data and transaction.
autoloMATE® provides a set of features to enhance Quality Assurance and Control such as special control tasks accessible only by qualified individuals and e-signing of the audit log review. Our software supports raising quality events, facilitating review and resolution.
By automating key parts of the execution process such as running calculations, transcribing data from batch records, forms & external systems like LIMS, autoloMATE® speeds up processes, eliminates errors & improves communication of data between key departments.
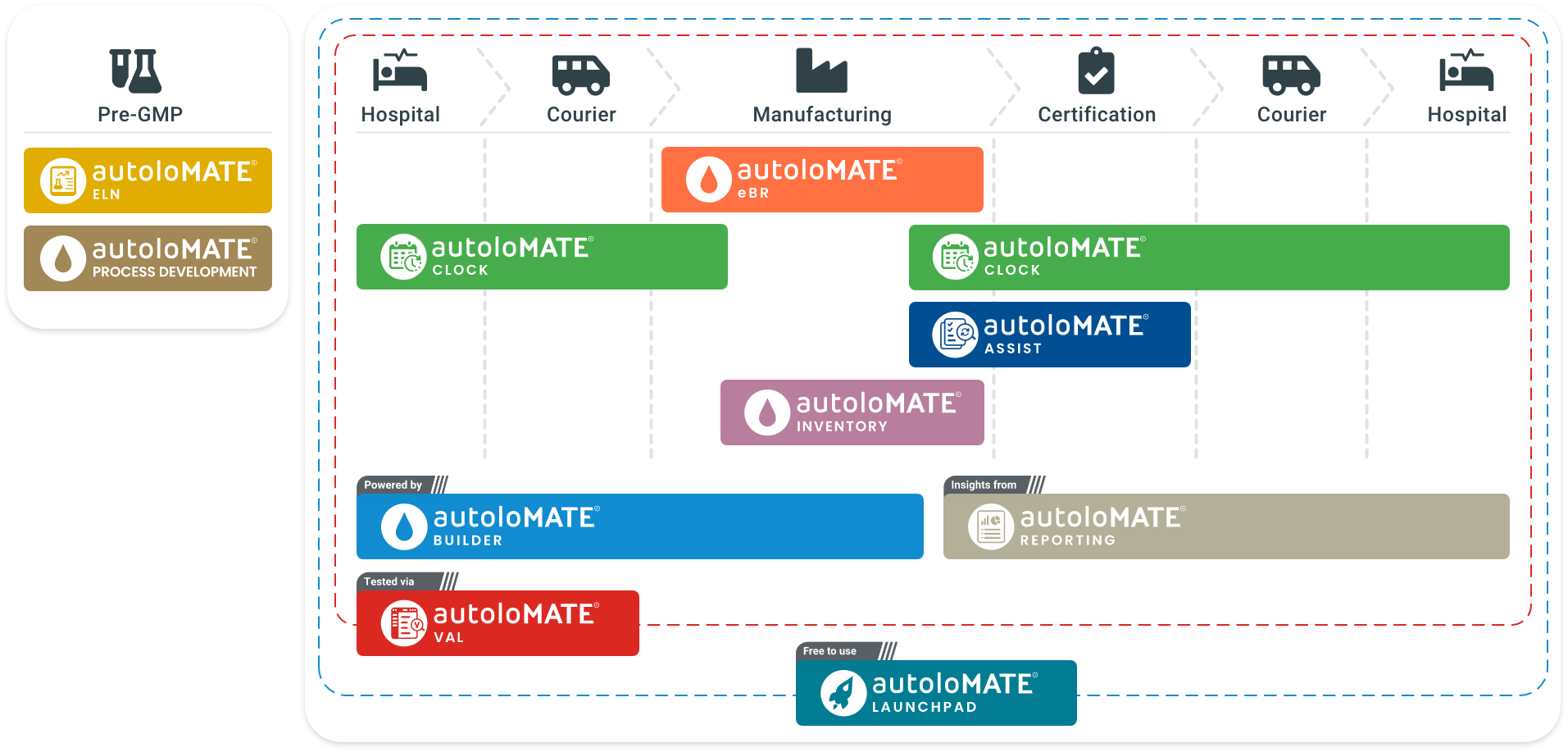
Autolomous provides organisations with ways to scale, by deploying a fully integrated, digitised and interoperable supply chain ecosystem.


autoloMATE® CLOCK digitises all aspects of scheduling to facilitate optimisation of facility operations. By accounting for all critical data — such as clinician schedules, equipment and clean room availability, and courier interactions — CLOCK enables scale-up and maximised utilisation of resources while driving down cost-of-goods.

autoloMATE® eBR digitises batch records, eliminating the need for vast amounts of paper. It has been designed to provide agility in a controlled environment, and can be configured for a wide range of life sciences workflows — from cell and gene therapy, including Autologous CAR-T, TIL, DC vaccine, and Allogeneic NK, iPSC and MSC, to broader biologics and pharmaceutical processes. Using real-time data verification, autoloMATE® eBR can accelerate the product review and release process, reducing the total time required from up to 36 hours to as little as 2.

autoloMATE Assist supports the batch verification of life sciences products. Data collated on the autoloMATE® platform via autoloMATE® eBR and autoloMATE® CLOCK will be assessed against specifications defined in the Critical Quality Attributes and visually presented to quality stakeholders (QC, QA, QP), enabling them to carry out assessments based on “review by exception” methodologies. Controlling and reducing manual data operations increases the integrity of the data, while reducing associated risks — together driving efficiency, throughput and the delivery of medicines and health products to patients.

Contact us If you have any questions about our Terms and Conditions, Privacy Policy or if you would…
08 Jun 2026
The autoloMATE® platform is a unified digital ecosystem that integrates a suite of specialized services to streamline life…
11 Feb 2026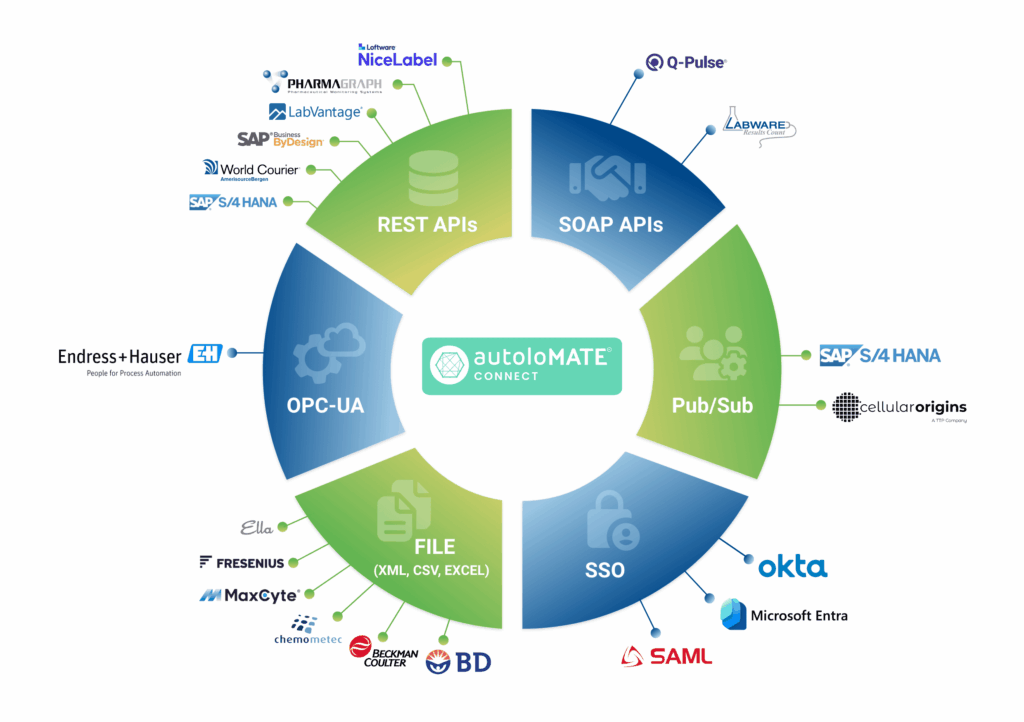
In the fast-evolving world of Cell and Gene Therapy (CGT), digital interoperability isn’t just a technical challenge —…
15 Jan 2026autoloMATE® comes with an always expanding set of integrations to external devices and systems, ranging from Laboratory Information Management Systems (LIMS) and Quality Management Systems (QMS) to cell counters & flow cytometers. These integrations allow us to get data from systems and devices and put them to use in the execution record, speeding up processes, eliminating transcription errors, and simplifying the use of version controlled documents such as standard operating procedures.
The eBR Builder allows users to configure and edit eBRs on the autoloMATE® platform on their own. It equips organisations with the resilience and agility required to adequately react to process changes while de-risking the time and cost of engaging the supplier.
The autoloMATE® Reporting module unlocks insights from within eBRs and forms, allowing for trends, bottlenecks and learnings to be identified and leveraged to achieve optimisation and scale-up.
Everything that happens within the autoloMATE® platform is tracked and available for review to our clients’ system administrators – from name changes to login / log out attempts, product records access, and more, providing an overall useful tool for investigative and record-keeping purposes.
User group segregation allows organisations such as CDMOs to keep data belonging to clients who use their facilities fully segregated while using a single platform. Groups can also be used to separate users into different projects within the same company.
Request more information
To delve further into what we do at Autolomous, contact us to request more information.
